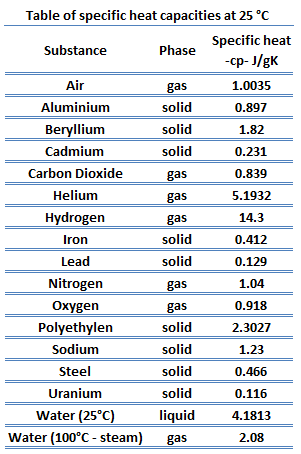
1. Calculate the amount of heat required to change the temperature of an iron ball of mass 3 kg from 30° C - Brainly.in

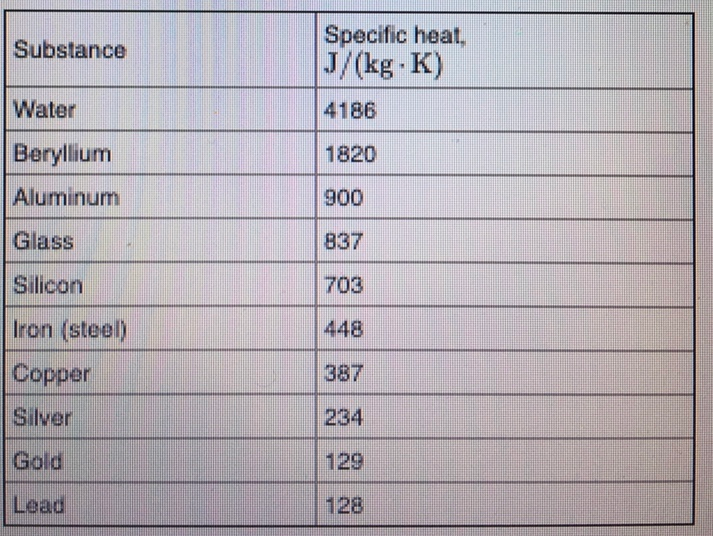
📐PSYW - Please Show Your Work A radiator made out of iron of specific heat capacity 450 J/kgk has a - Brainly.com
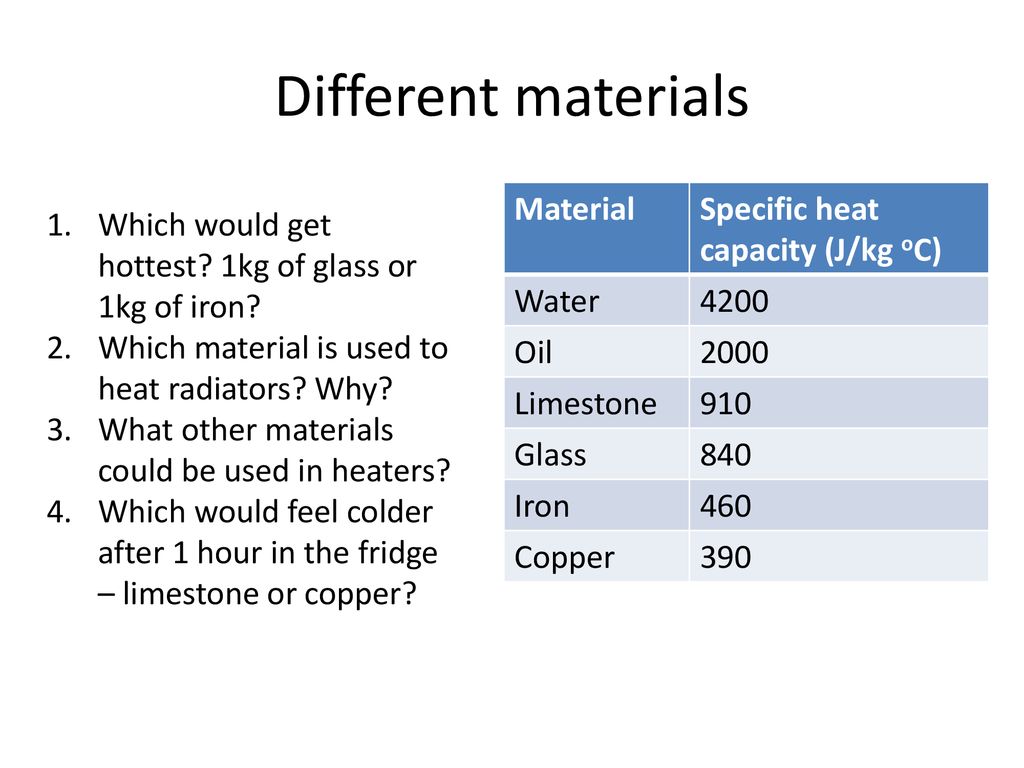
Learning Outcomes: Rearranging equation for Specific Heat Capacity Topic Equation for Specific Heat Capacity Target Audience: G & T Teacher instructions. - ppt download

SOLVED: (IV) The specific heat capacity of aluminium is CAt 900 J /(kg-C) , while that of iron is CFe 450 J/(kg-C). (a) Five kilograms of aluminium at 20€ and 10kg ol




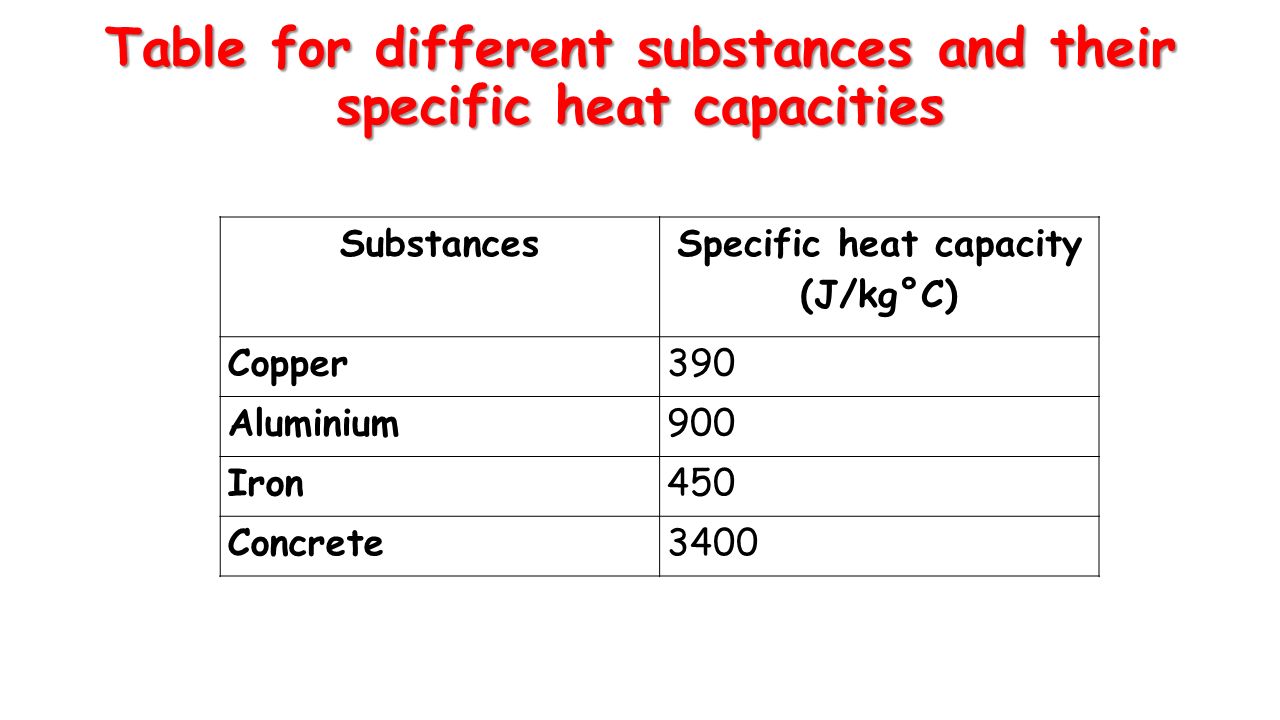
![tv and Lv of water and some metals [13] | Download Scientific Diagram tv and Lv of water and some metals [13] | Download Scientific Diagram](https://www.researchgate.net/publication/341481178/figure/tbl1/AS:892847209316355@1589882771919/tv-and-Lv-of-water-and-some-metals-13.png)