![Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download](https://images.slideplayer.com/15/4572254/slides/slide_3.jpg)
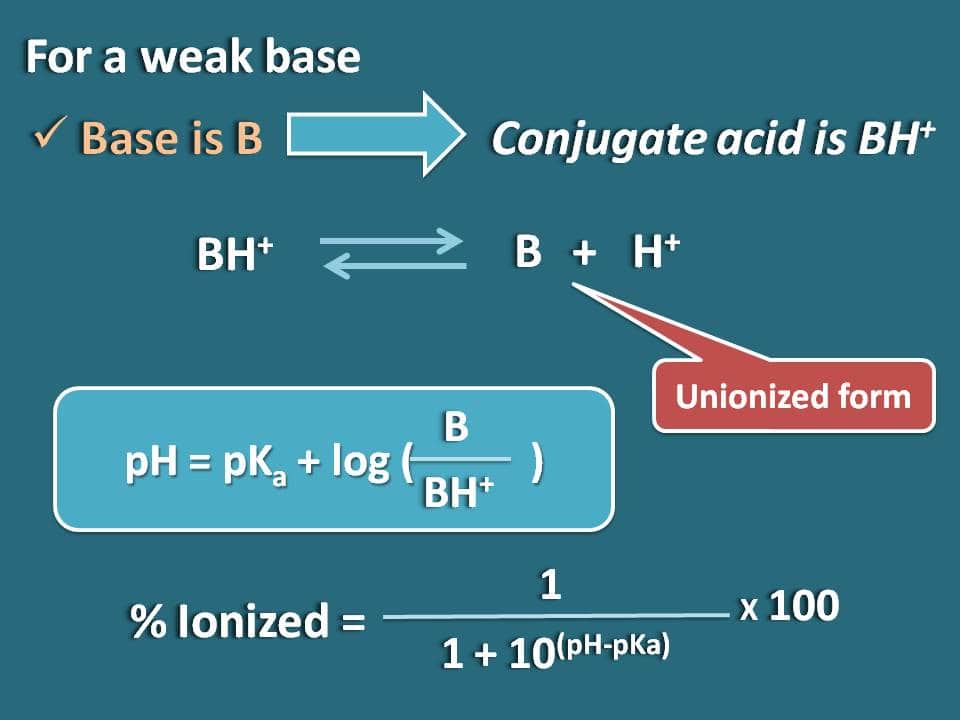
Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download
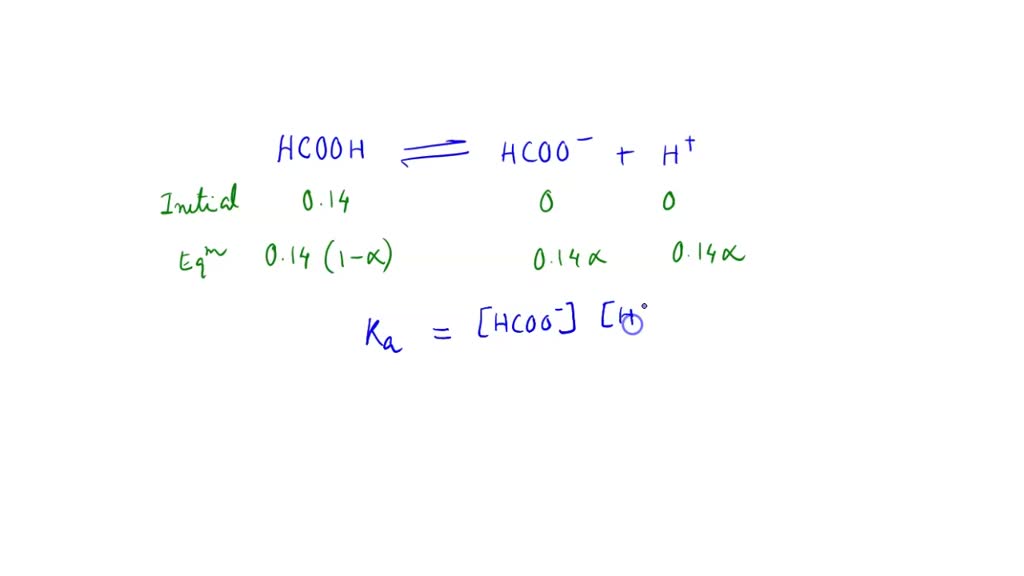
SOLVED: Calculate the percent ionization of hippuric acid, C9H9NO3 (Ka = 2.4 x 10-4), in a 0.174 M aqueous solution. Report your answer to one decimal place.

An example species diagram showing the percent ionization of polymyxin... | Download Scientific Diagram

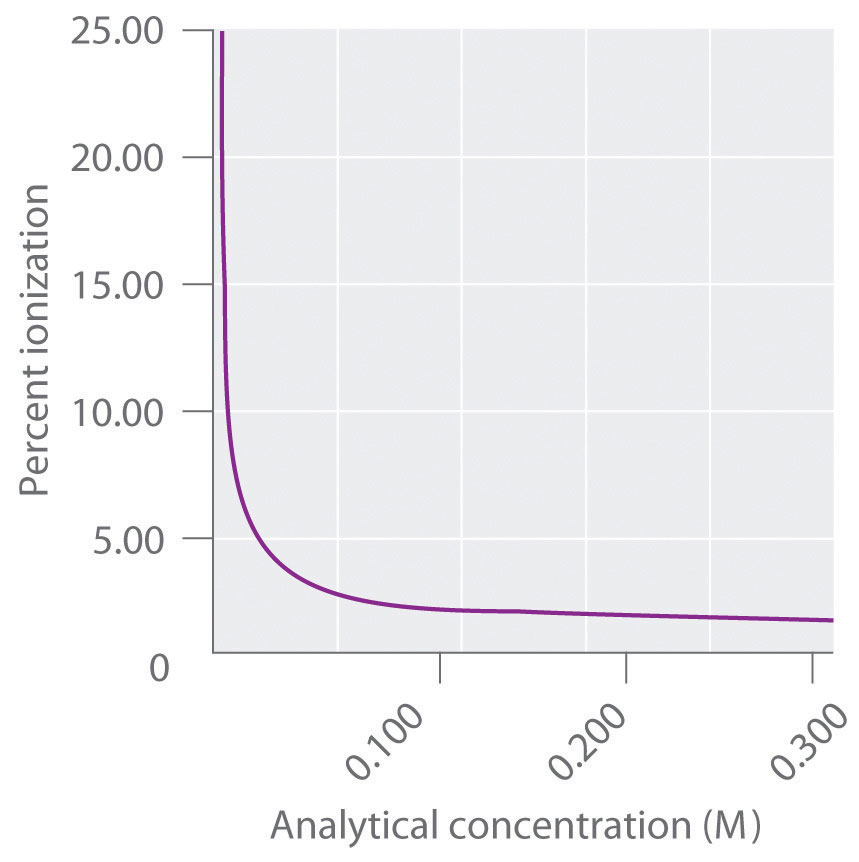
Figure 1. Plot of degree of ionization () versus pH : Do We Really Need the Concentration and Volume of a Solution of a Weak Acid to Calculate the Percentage Ionization or

Worked example: Finding the percent ionization of a weak acid | AP Chemistry | Khan Academy - YouTube
![Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download](https://slideplayer.com/4572254/15/images/slide_1.jpg)