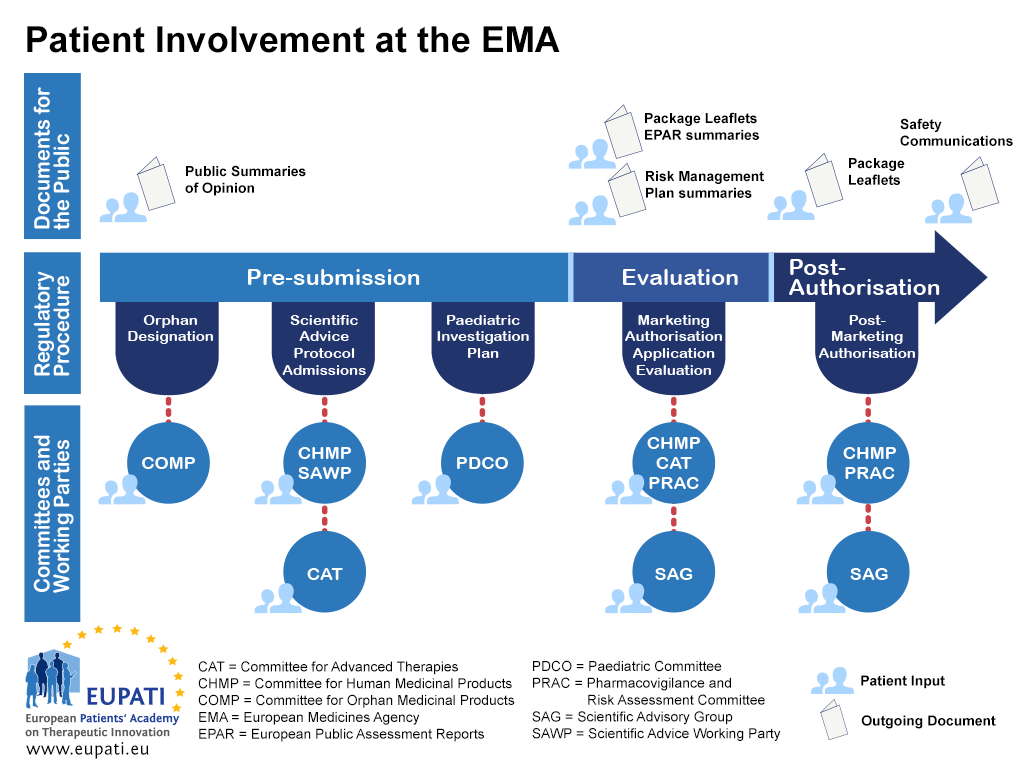
EU approved biosimilars are interchangeable with reference medicines/ equivalent biosimilar: EMA, HMA, Health News, ET HealthWorld

European Medicines Agency (EMA) launches pilot project on drug repurposing – EJP RD – European Joint Programme on Rare Diseases

Flowchart of included drugs. EMA, European Medicines Agency; FDA, Food... | Download Scientific Diagram

EMA is launching a pilot to support the repurposing of established medicines for use in new indications by NGOs and academia. | ERICA

EU Medicines Agency on Twitter: "EMA's safety committee, the #PRAC, starts review of #terlipressin medicines and recommends updates to product information for some #COVID19vaccines: 👉https://t.co/EFGDJII0Xt #SafetyOfVaccines https://t.co/wLcVnqYMDs ...











.jpg)