
Late-Stage Carbon-14 Labeling and Isotope Exchange: Emerging Opportunities and Future Challenges | JACS Au

C-11 is a radioisotope of carbon. Its half - life is 20 min. What fraction of the initial number of C-11 atoms in a sample will have decayed away after 80 min ?
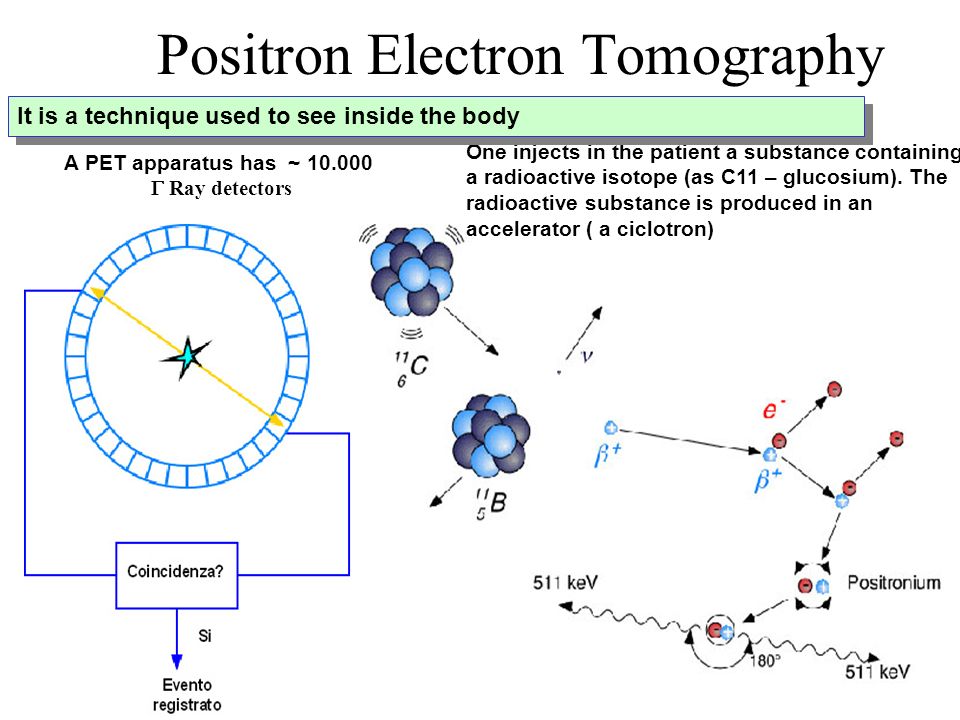
1. Positron Electron Tomography One injects in the patient a substance containing a radioactive isotope (as C11 – glucosium). The radioactive substance. - ppt download
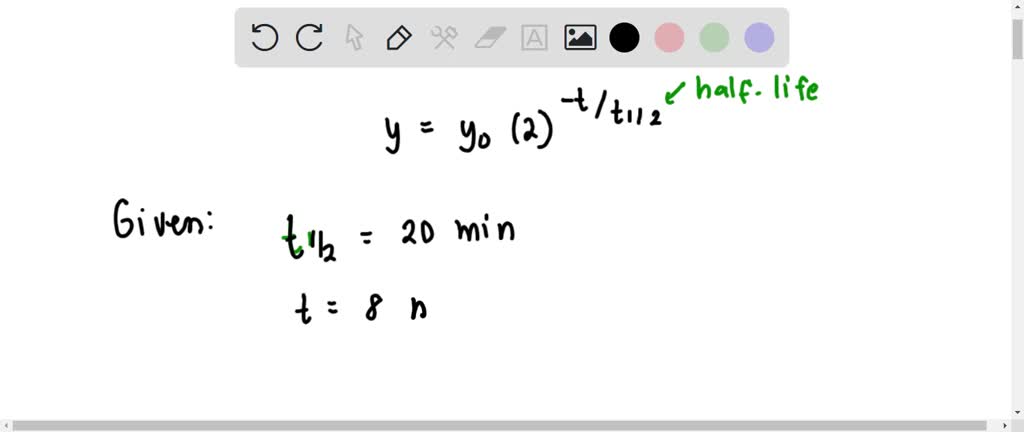
SOLVED: Carbon-11 is a radioactive isotope of carbon. Its half-life is 20 minutes. What fraction of the initial number of C-11 atoms in a sample will have decayed away after 80 minutes?
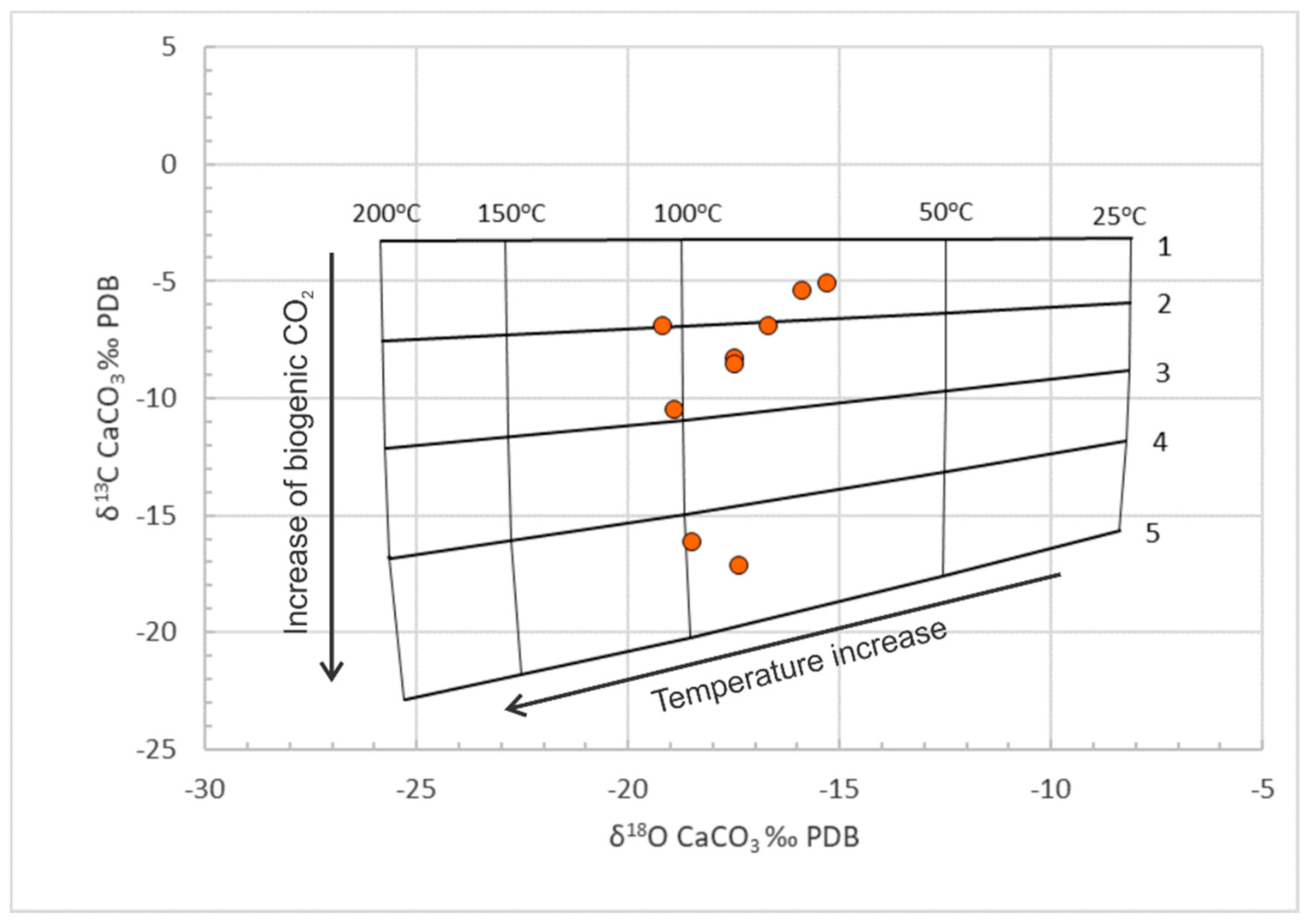
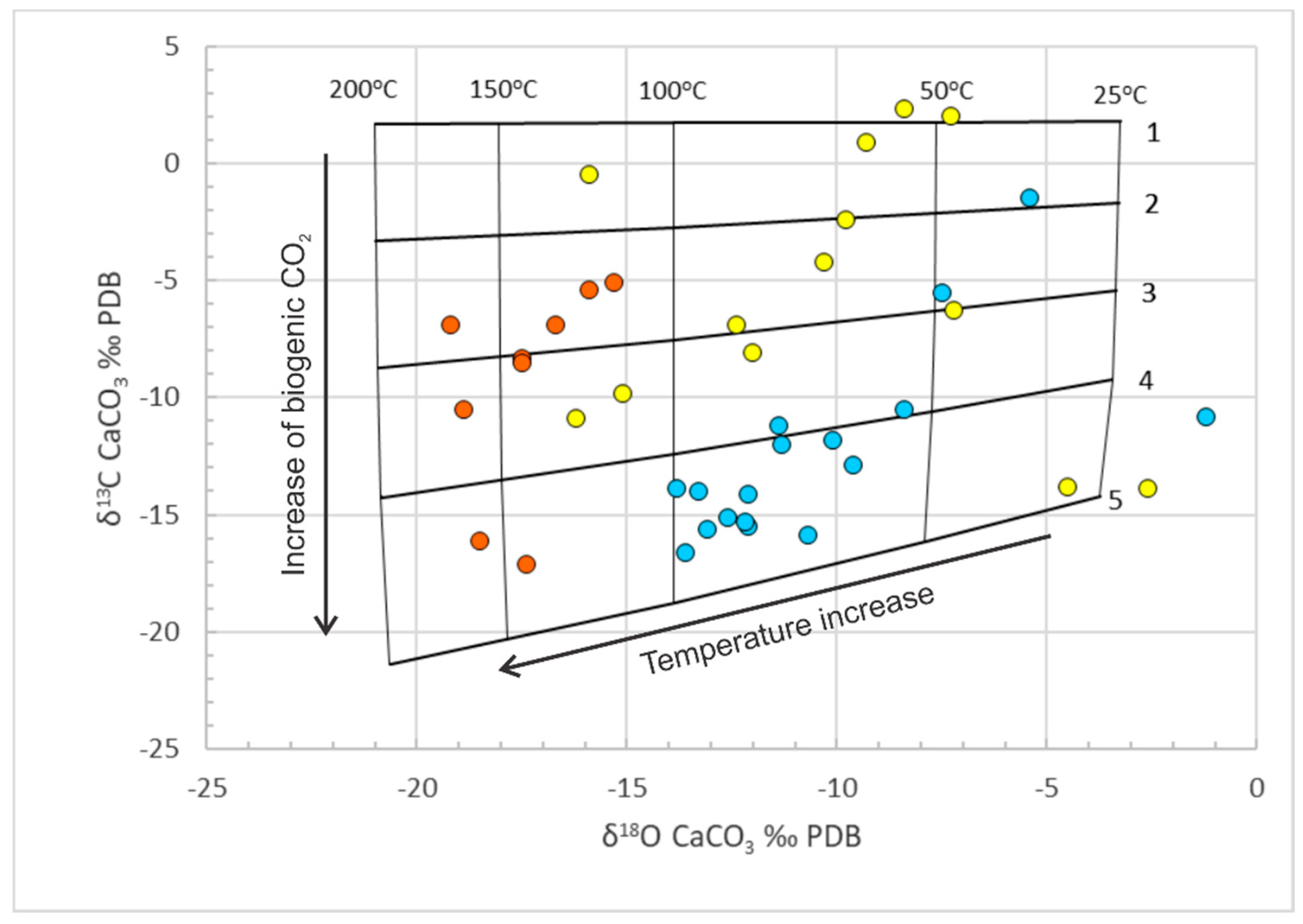
Geosciences | Free Full-Text | Application of the Data on δ13C and δ18O of Carbonates for the Study of Unconventional Reservoirs on the Example of the Bazhenov Source Rocks, Western Siberia, Russia
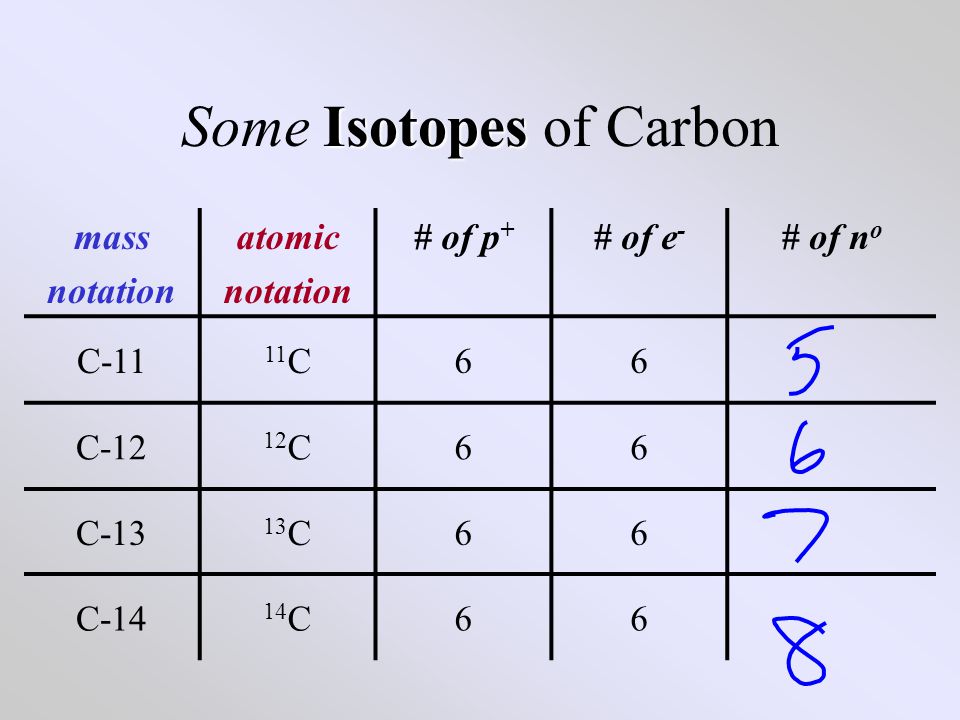
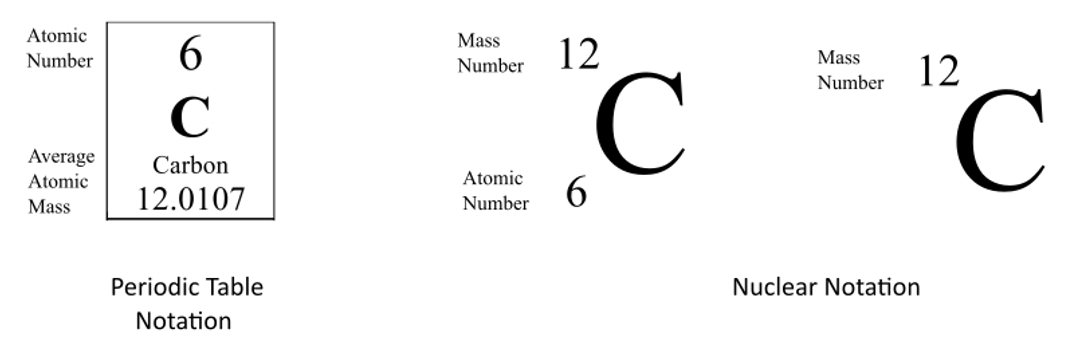
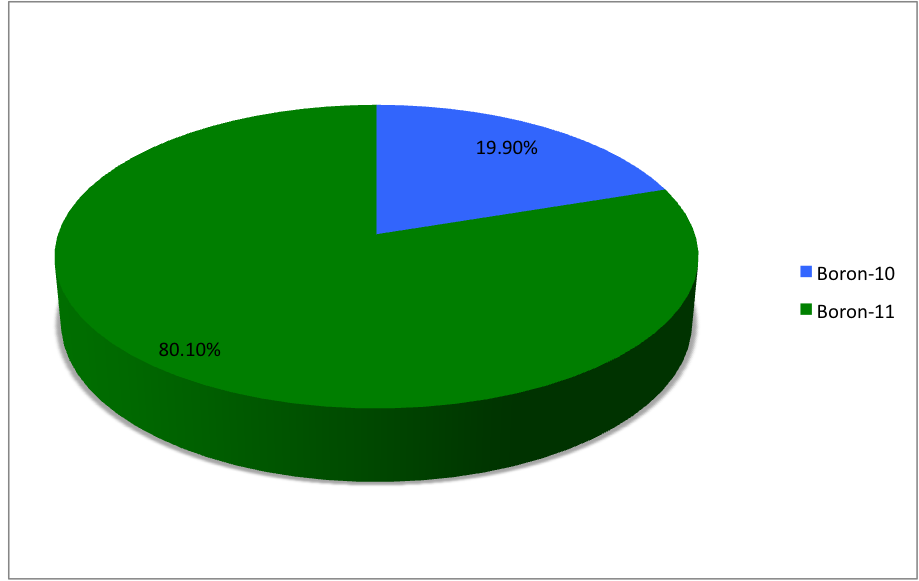
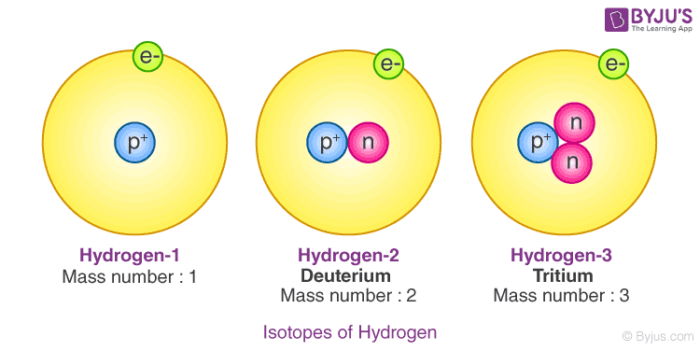
Isotopes atoms of a given element that differ in the number of neutrons …and consequently in mass. - ppt download








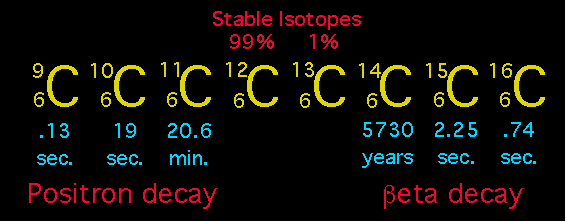





![Efficiency of [ 13 C 11 15 N 2 ]Trp stable isotope incorporation into IAA | Download Table Efficiency of [ 13 C 11 15 N 2 ]Trp stable isotope incorporation into IAA | Download Table](https://www.researchgate.net/publication/38074803/figure/tbl1/AS:394274555351040@1471013787138/Efficiency-of-13-C-11-15-N-2-Trp-stable-isotope-incorporation-into-IAA.png)